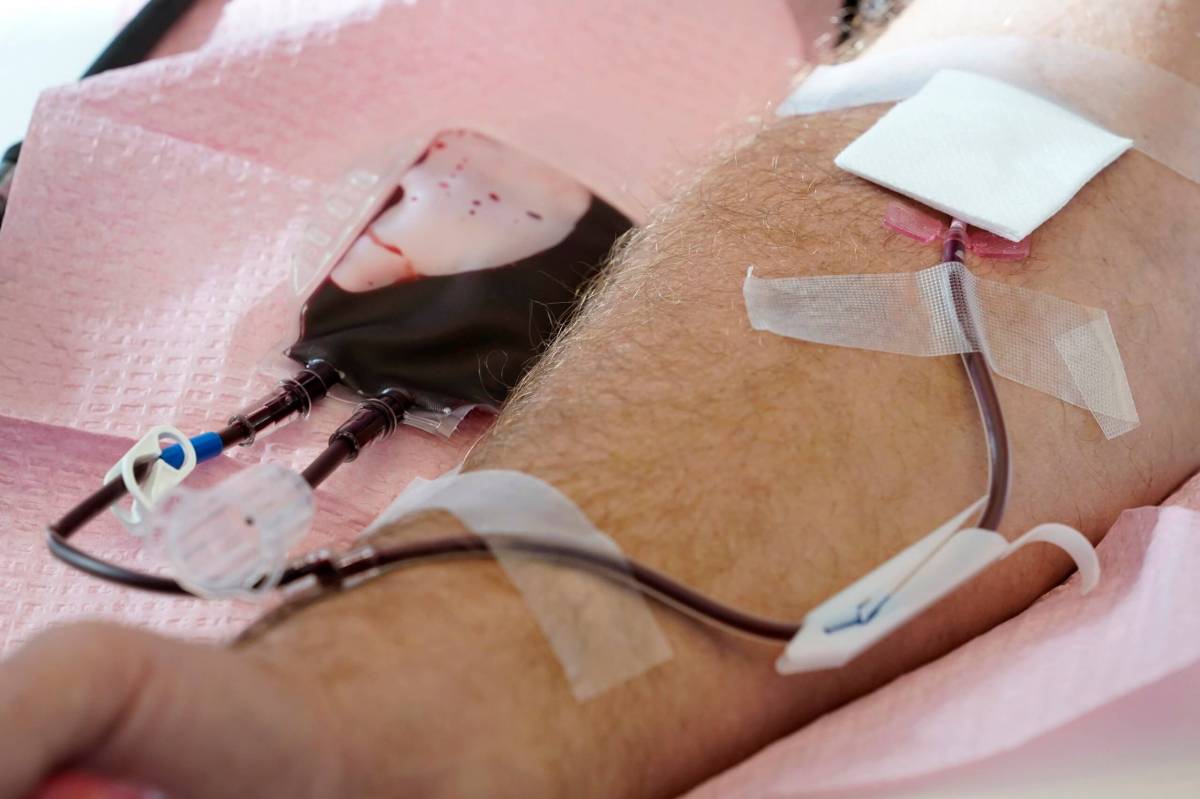
The U.S. is moving to further ease restrictions on blood donations from gay and bisexual men and other groups that typically face higher risks of HIV.
The Food and Drug Administration on Friday announced draft guidelines that would do away with the current three-month abstinence requirement for donations from men who have sex with men. Instead, all potential donors would be screened with a new questionnaire that evaluates their individual risks for HIV based on sexual behavior, recent partners and other factors.
If finalized, many gay and bisexual men in monogamous relationships would be able to donate blood for the first time in decades. It’s the latest move by the FDA to broaden donor eligibility, with the potential to boost donations.
“We feel confident that the safety of the blood supply will be maintained,” FDA’s Dr. Peter Marks told reporters.
Gay rights groups have long opposed blanket restrictions on who can give blood, saying they discriminate against the LGBTQ community. Medical societies including the American Medical Association have also said such exclusions are unnecessary given advances in technology to test blood for infectious diseases.
“Current and former blood donation policies made unfounded assumptions about gay and bisexual men and really entangled individuals’ identity with their likelihood of having HIV,” said Sarah Warbelow of the Human Rights Campaign, an LGBTQ advocacy group.
The U.S. and many other countries started blocking blood donations from gay and bisexual men during the early 1980’s AIDS epidemic, aiming to prevent the spread of HIV through the blood supply.
In 2015, the FDA dropped the lifetime ban and replaced it with a one-year abstinence requirement. Then in 2020, the agency shortened the abstinence period to three months, after donations plummeted during the COVID-19 pandemic.
Regulators said there has been no negative impact on the blood supply as a results of those changes.
The FDA sets requirements and procedures for blood banks throughout the U.S. All potential donors answer questions about their sexual history, injectable drug use and any recent tattoos or piercing, among other factors that can contribute to the spread of blood-borne infections. Donated blood is then tested for HIV, hepatitis C, syphilis and other infectious diseases.
Under the new proposal, men who have sex with men will be asked if they have had new or multiple partners in the last three months. Those who answer affirmatively to either question and also report having anal sex would be barred from donating until a later date. The policy would also apply to women who have sex with gay or bisexual men.
Anyone who has ever tested positive for HIV would continue to be ineligible to donate blood. Those taking pills to prevent HIV through sexual contact would also still be barred, until three months after their last dose. The FDA noted that the medication, known as PrEP, can delay the detection of the virus in screening tests.
Marks said the agency is willing to consider further easing restrictions “but we have to have the science to do that.”
FDA regulators will take public comments on the proposal for 60 days before beginning to finalize the guidelines.
The proposed policy mirrors those used in Canada and the U.K.
LGBTQ groups welcomed the FDA’s announcement but said the proposal should not exclude people using PrEP medications.
“We must be conscious to not further stigmatize these safe sex practices and uplift individuals taking precautions,” said Jose Abrigo of Lambda Legal, which has long pushed to change the FDA policy.
The FDA based its latest proposal, in part, on a recent study of 1,600 gay and bisexual men. The FDA-funded research compared the effectiveness of a detailed, personalized questionnaire on sexual behavior to the current time-based abstinence rules.
It will take several months for blood banks to make the changes, according to Cliff Numark, an executive with Vitalant, a blood center that participated in the study. The changes will require new questionnaires, training for staff and updating computer software.
The Red Cross said it supports the FDA changes but added that it’s too early to know if they will result in more blood donations.
Lukas Pietrzak of Washington D.C., said he eagerly volunteered for the FDA study. He credits emergency blood transfusions with saving his father’s life after a cycling accident in 1991.
Pietrzak donated blood in high school but became ineligible after becoming sexually active as a gay man.
“Until I fully came out to my friends, I had to skirt around why I never went to blood drives with them,” says Pietrzak, 26, who now works for the federal government.
When there are calls for blood donations “now we’re able to be part of that,” Pietrzak said.
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Science and Educational Media Group. The AP is solely responsible for all content.